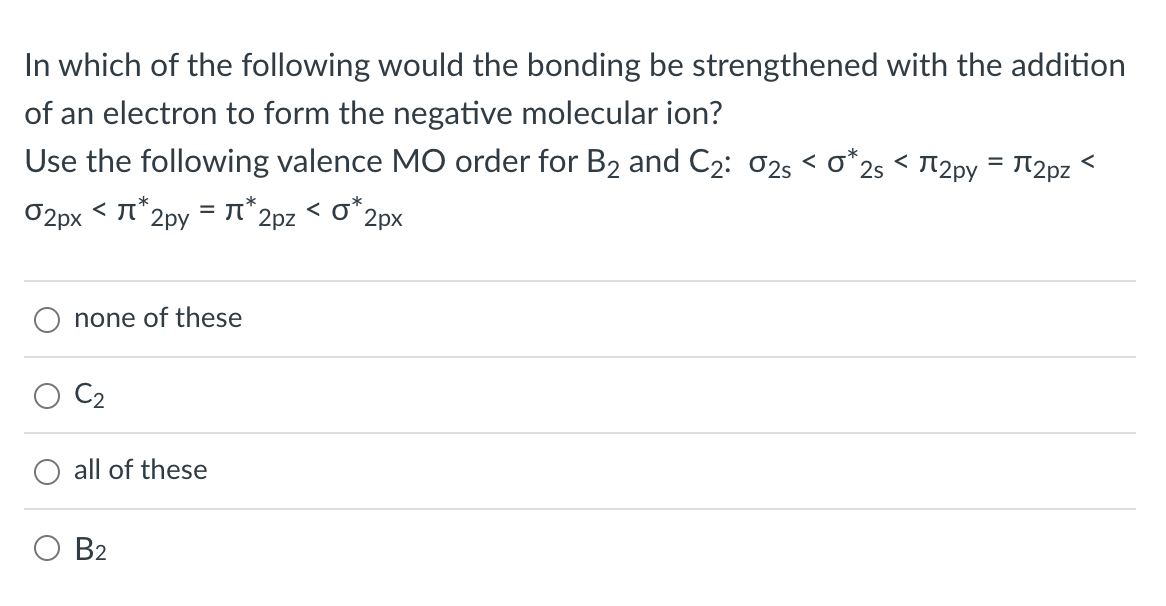Home /
Expert Answers /
Chemistry /
the-following-images-show-lewis-structures-containing-hypothetical-atoms-a-x-and-e-with-di-pa758
(Solved): The following images show Lewis structures containing hypothetical atoms A, \( X \), and E with di ...
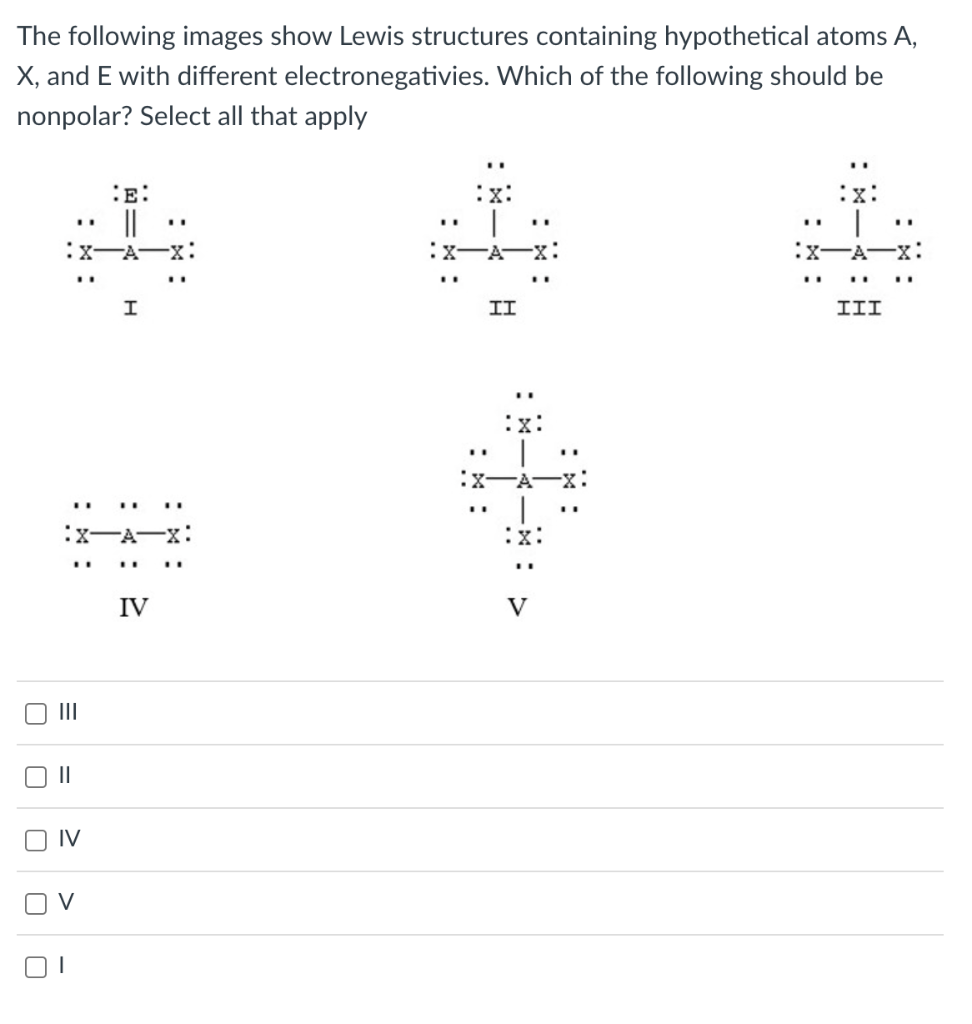
The following images show Lewis structures containing hypothetical atoms A, \( X \), and E with different electronegativies. Which of the following should be nonpolar? Select all that apply III II IV V
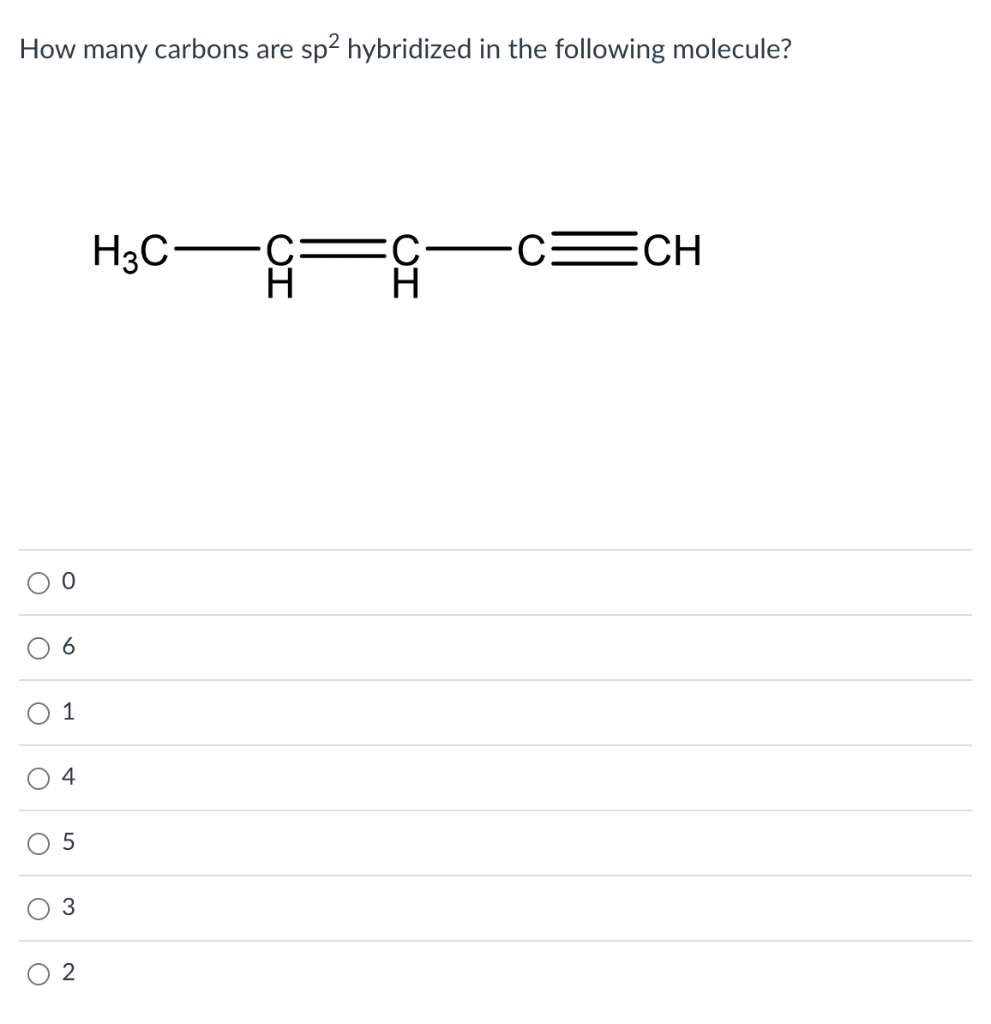
How many carbons are \( \mathrm{sp}^{2} \) hybridized in the following molecule? 0 6 1 4
In which of the following would the bonding be strengthened with the addition of an electron to form the negative molecular ion? Use the following valence \( \mathrm{MO} \) order for \( \mathrm{B}_{2} \) and \( \mathrm{C}_{2}: \sigma_{2 \mathrm{~s}}<\sigma_{2 \mathrm{~s}}^{*}<\pi_{2 \mathrm{py}}=\pi_{2 \mathrm{pz}}< \) \( \sigma_{2 p x}<\pi^{*} 2 p y=\pi^{*} 2 p z<\sigma_{2 p x}^{*} \) none of these \( \mathrm{C}_{2} \) all of these \( B_{2} \)
Expert Answer
Molecule in Structure 1 is polar