Home /
Expert Answers /
Chemistry /
predict-how-chemical-equilbria-will-change-with-changes-in-temperature-pressure-and-concertration-pa290
(Solved): Predict how chemical equilbria will change with changes in temperature, pressure, and concertration. ...
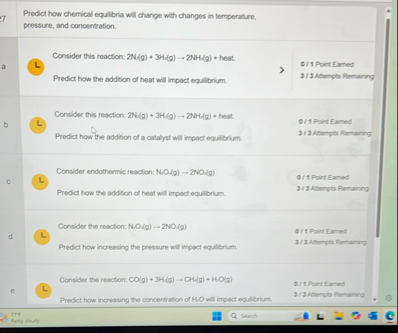
Predict how chemical equilbria will change with changes in temperature, pressure, and concertration. Consider this reaction:
2N(g) 3(H)/(g)-2NH(g) heat. 0/1 Port Eamed Predict how the addition of heat will impact equilbrium. 1/12Bempts Remaing Consider this reaction:
2N-(g) 3H(g)->2NH(g) heat. 0/1 Poirt Eamed Predict how the addition of a catalyst will impact equiliorium. 1/3ABerpts Remany c Consider endothermic reaction:
N*O(g)-2NO(g)0/1 Point Earned Prodict how the addition of heat will impact equilbrium. 2/3ASempts Remain y Consider the resction:
N.O(q)->2NO(q)0/14 Poirt Eamed Predict how incressing the pressure will Impact equilibrium. 1/3 ASerges Remarn: e Consider the rewction: 0/1 Poirt Eaned Predict how increasing tie concentration of Ht 2/2Atencts Remainy IT Hesech hetly doudy