Home /
Expert Answers /
Chemistry /
part-1-provide-the-bond-angles-a-e-in-the-following-molecules-circle-all-the-sp2-hybridized-carbon-pa603
(Solved): Part 1 Provide the bond angles a-e in the following molecules. Circle all the sp2 hybridized carbon ...
Part 1
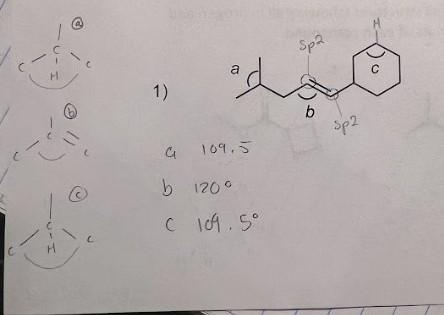
Provide the bond angles a-e in the following molecules. Circle all the sp2 hybridized carbons and box all the sp hybridized carbons.
- I don't understand how we got 109.5 degrees and did I circle everything correctly?
Part 2
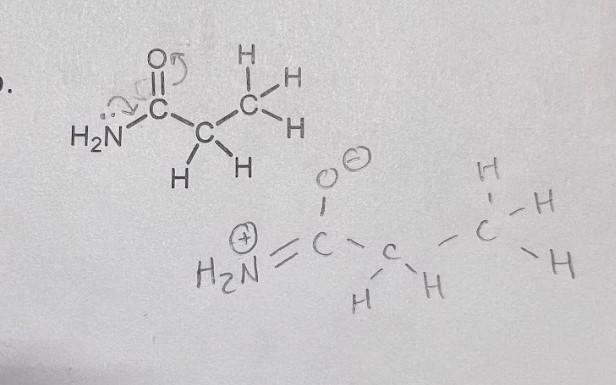
I also don't understand where the lone pairs came from on the H2N when they weren't originally there on the initial resonance structure? Why can't we push the double bond onto the carbon and then push that onto the bond between carbon and H2N to make that a double bond?
1) a b C
Expert Answer
Part 1carbon labeled with a is sp3 hybridized in natu