Home /
Expert Answers /
Chemistry /
in-radical-chlorination-of-alkanes-nonequivalent-hydrogens-react-with-chlorine-atoms-at-different-pa792
(Solved): In radical chlorination of alkanes, nonequivalent hydrogens react with chlorine atoms at different ...

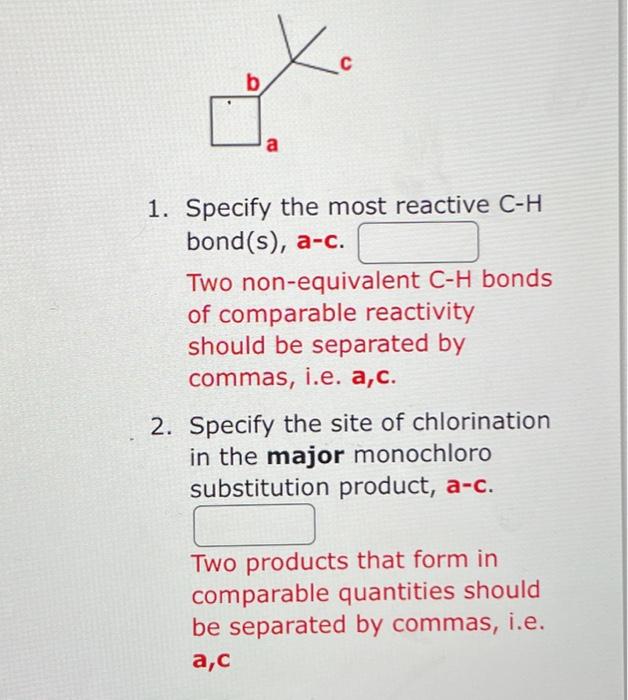

In radical chlorination of alkanes, nonequivalent hydrogens react with chlorine atoms at different rates. At , primary, secondary, and tertiary bonds react at relative rates of : 5.2 respectively. These are conditions of kinetic control where product ratios are determined by relative rates of formation. For example, if is formed twice as fast as , the product ratio will be 2 . Consider chlorination of the alkane below at . 1. Specify the most reactive bond(s), a-c. Two non-equivalent bonds of comparable reactivity should be separated by commas, i.e. a,c. 2. Specify the site of chlorination in the major monochloro substitution product, a-c. Two products that form in comparable quantities should be separated by commas, i.e.
1. Specify the most reactive bond(s), a-c. Two non-equivalent bonds of comparable reactivity should be separated by commas, i.e. a,c. 2. Specify the site of chlorination in the major monochloro substitution product, a-c. Two products that form in comparable quantities should be separated by commas, i.e.
1. Specify the most reactive bond(s), a-c. Two non-equivalent bonds of comparable reactivity should be separated by commas, i.e. a,c. 2. Specify the site of chlorination in the major monochloro substitution product, a-c. Two products that form in comparable quantities should be separated by commas, i.e.
Expert Answer
In radical chlorination of