(Solved): Functional Groups in Organic Molecules In the activity below you will determine the chemical formul ...
Functional Groups in Organic Molecules
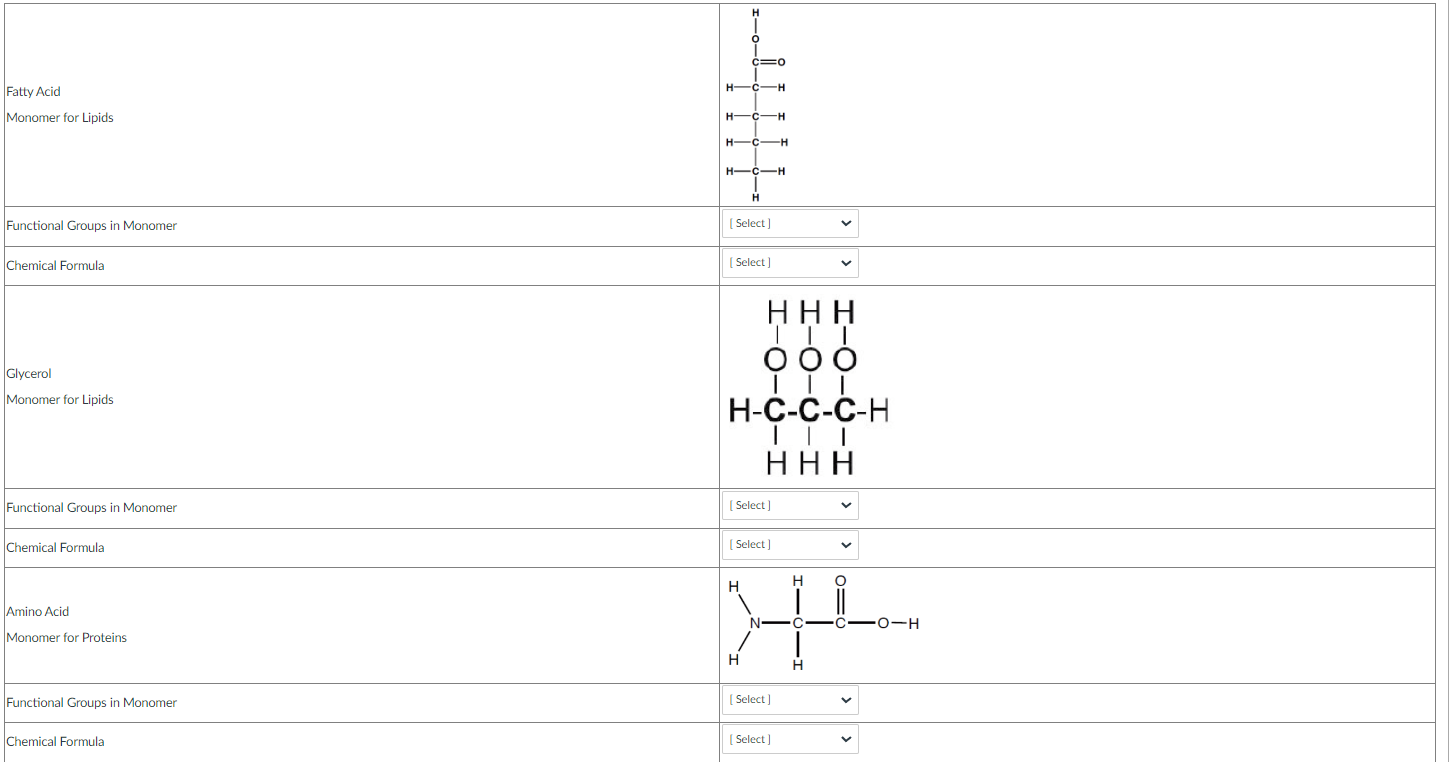
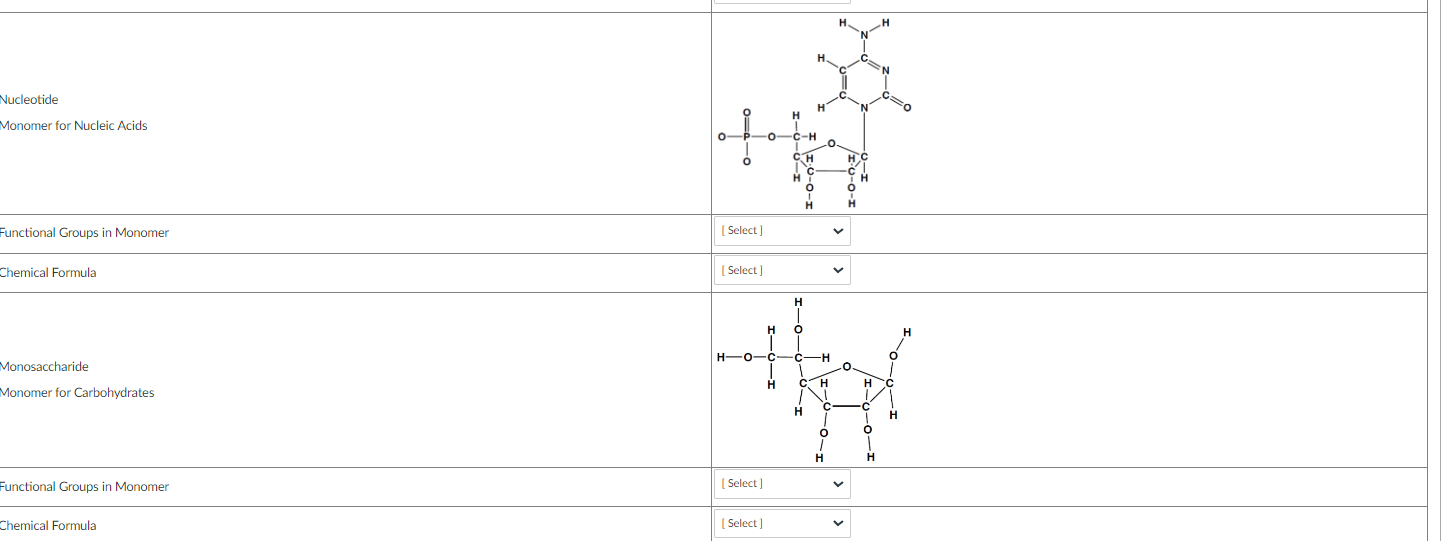
In the activity below you will determine the chemical formula and identify the functional groups in the following monomers for biological molecules. Some of the monomers will have more than one functional group attached to the carbon skeleton. To determine the chemical formula you will count the number of each element and use this number with the element symbol to make the chemical formula.
Here is an example that is worked out for you to help you get started:
You can see that this molecule of propionic acid that there is 1 circled carboxyl functional group.
Also the propionic acid molecule has 3 Carbons (C3), 6 Hydrogens (H6), and 2 Oxygens (O2) giving it a chemical formula of C3H6O2.
Using the information above choose the correct answers in the table below.
*Please note that the chemical formula format is limited in these types of questions. Read the chemical formulas carefully.