Home /
Expert Answers /
Chemistry /
determine-the-molecular-formula-for-the-compound-with-a-molar-mass-of-88-16-mathrm-g-mathr-pa426
(Solved): Determine the molecular formula for the compound with a molar mass of \( 88.16 \mathrm{~g} / \mathr ...
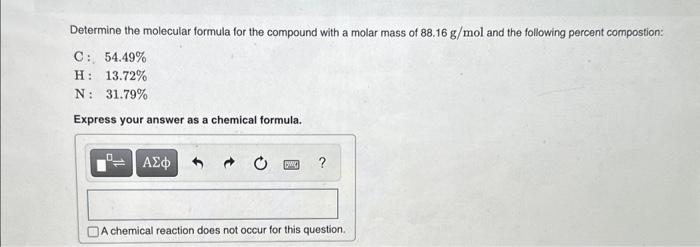
Determine the molecular formula for the compound with a molar mass of \( 88.16 \mathrm{~g} / \mathrm{mol} \) and the following percent compostion: \[ \begin{array}{ll} \mathrm{C}: & 54.49 \% \\ \mathrm{H}: & 13.72 \% \\ \mathrm{~N}: & 31.79 \% \end{array} \] Express your answer as a chemical formula.