Home /
Expert Answers /
Chemistry /
consider-each-pair-of-structural-formulas-that-follow-and-state-whether-the-two-formulas-represent-pa420
(Solved): Consider each pair of structural formulas that follow and state whether the two formulas represent ...
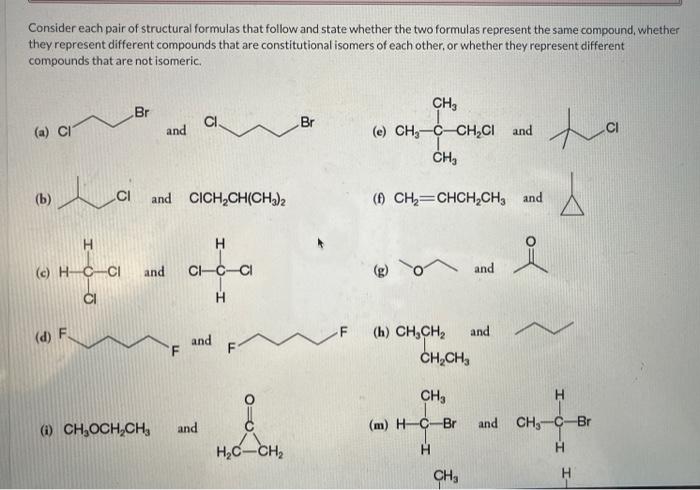
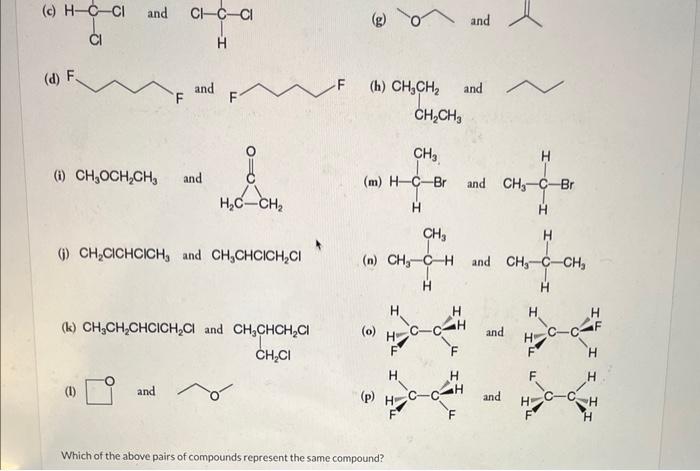



Consider each pair of structural formulas that follow and state whether the two formulas represent the same compound, whether they represent different compounds that are constitutional isomers of each other, or whether they represent different compounds that are not isomeric. (a) (e) (b) (f) \( \mathrm{CH}_{2}=\mathrm{CHCH}_{2} \mathrm{CH}_{3} \) and (c) and (g) (d) (h) \( \mathrm{CH}_{3} \mathrm{CH}_{2} \) and (i) \( \mathrm{CH}_{3} \mathrm{OCH}_{2} \mathrm{CH}_{3} \) and (m) and
(h) \( \mathrm{CH}_{3} \mathrm{CH}_{2} \) and (i) \( \mathrm{CH}_{3} \mathrm{OCH}_{2} \mathrm{CH}_{3} \) and (m) (j) \( \mathrm{CH}_{2} \mathrm{ClCHClCH} \) and \( \mathrm{CH}_{3} \mathrm{CHClCH}_{2} \mathrm{Cl} \) (n) (k) \( \mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CHClCH}_{2} \mathrm{Cl} \) and (I) and Which of the above pairs of compounds represent the same compound?
Which of the above pairs of compounds represent the same compound? A B C D E F G H I K L M N p
Which of the above pairs of compounds represent constitutional isomers?
Which of the above pairs of compounds represent different compounds, not isomeric?