Home /
Expert Answers /
Chemistry /
at-1-bar-how-much-energy-is-required-to-heat-63-0-mathrm-g-mathrm-h-2-mathrm-o-mathrm-pa885
(Solved): At 1 bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H}_{2} \mathrm{O}(\mathrm ...
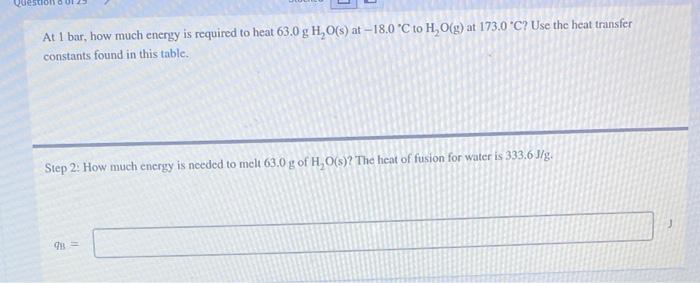



At 1 bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H}_{2} \mathrm{O}(\mathrm{s}) \) at \( -18.0^{\circ} \mathrm{C} \) to \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) at \( 173.0^{\circ} \mathrm{C} \) ? Use the heat transfer constants found in this table. Step 2: How much energy is needed to melt \( 63.0 \mathrm{~g} \mathrm{of} \mathrm{H}_{2} \mathrm{O}(\mathrm{s}) \) ? The heat of fusion for water is \( 333.6 \mathrm{~J} / \mathrm{g} \).
At I bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H} \mathrm{O}(\mathrm{s}) \) at \( -18.0^{\circ} \mathrm{C} \) to \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) at \( 173.0^{\circ} \mathrm{C} \) ? Use the heat transfer constants found in this table. Step 3: How much energy is needed to heat \( 63.0 \mathrm{~g} \mathrm{H}_{2} \mathrm{O}(\mathrm{I}) \) from \( 0.0^{\circ} \mathrm{C} \) to \( 100.0 \) " \( \mathrm{C} \) ? The specific heat of \( \mathrm{H}_{2} \mathrm{O}(1) \) is \( 4.184 \mathrm{~J} /(\mathrm{g} \cdot \mathrm{K}) \).
At 1 bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H} \mathrm{H}_{2} \mathrm{O}(\mathrm{s}) \) at \( -18.0^{\circ} \mathrm{C} \) to \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) at \( 173.0^{\circ} \mathrm{C} \) ? Use the heat transfer constants found in this table. Step 4: How much energy is needed to boil \( 63.0 \mathrm{~g} \mathrm{of} \mathrm{H}_{2} \mathrm{O}(\mathrm{l}) \) ? The heat of vaporization for water is \( 2257 \mathrm{~J} / \mathrm{g} \).
At 1 bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H} \mathrm{O}(\mathrm{s}) \) at \( -18.0^{\circ} \mathrm{C} \) to \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) at \( 173.0{ }^{\circ} \mathrm{C} \) ? Use the heat transfer constants found in this tablc. Step 5: How much energy is needed to heat \( 63.0 \mathrm{~g} \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) from \( 100.0^{\circ} \mathrm{C} \) to \( 173.0^{\circ} \mathrm{C} \) ? The specific heat of \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) is \( 2.000 \mathrm{~J} /(\mathrm{g} \cdot \mathrm{K}) \)
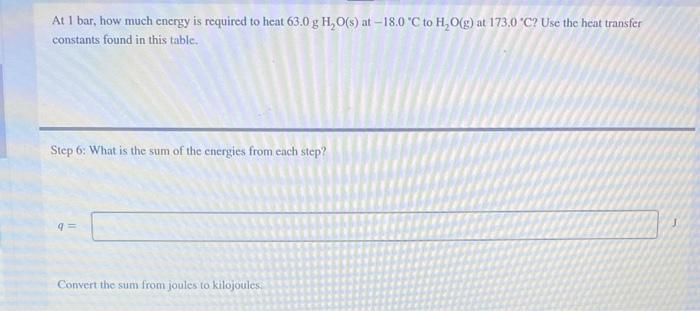
At 1 bar, how much energy is required to heat \( 63.0 \mathrm{~g} \mathrm{H}_{2} \mathrm{O}(\mathrm{s}) \) at \( -18.0^{\circ} \mathrm{C} \) to \( \mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \) at \( 173.0{ }^{\circ} \mathrm{C} \) ? Use the heat transfer constants found in this table. Step 6: What is the sum of the energies from each step? Convert the sum from joules to kilojoules:
Expert Answer
Given - Mass of water (m) = 63.0 g Initial temperature = -18.0 oC Final temperature = 173.0 oC Specific heat of H2O(s) = 2.1 J/g.K = 2.1 J/g.oC The heat of fusion () = 333.6 J/g Specific heat of H2O(l) = 4.184 J/g.K = 4.184 J/g.oC The heat of vaporiz