Home /
Expert Answers /
Chemistry /
a-buffered-solution-containing-dissolved-aniline-c-6-h-5-nh-2-and-aniline-hydrochloride-c-6-pa412
(Solved): A buffered solution containing dissolved aniline, C_(6)H_(5)NH_(2), and aniline hydrochloride, C_(6) ...
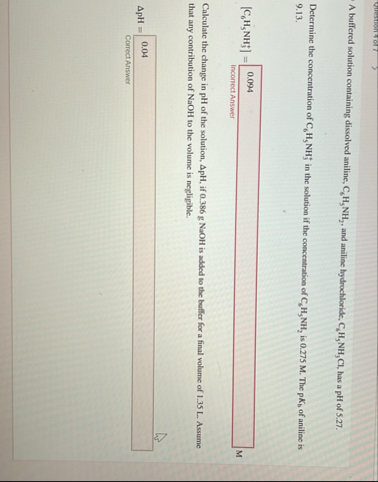
A buffered solution containing dissolved aniline,
C_(6)H_(5)NH_(2), and aniline hydrochloride,
C_(6)H_(5)NH_(3)Cl, has a pH of 5.27 . Determine the concentration of
C_(6)H_(5)NH_(3)^( )in the solution if the concentration of
C_(6)H_(5)NH_(2)is 0.275 M . The
pK_(b)of aniline is 9.13. Calculate the change in pH of the solution,
\Delta pH, if 0.386 g NaOH is added to the buffer for a final volume of 1.35 L . Assume that any contribution of NaOH to the volume is negligible.
\Delta pH=
◻Conecimiswas